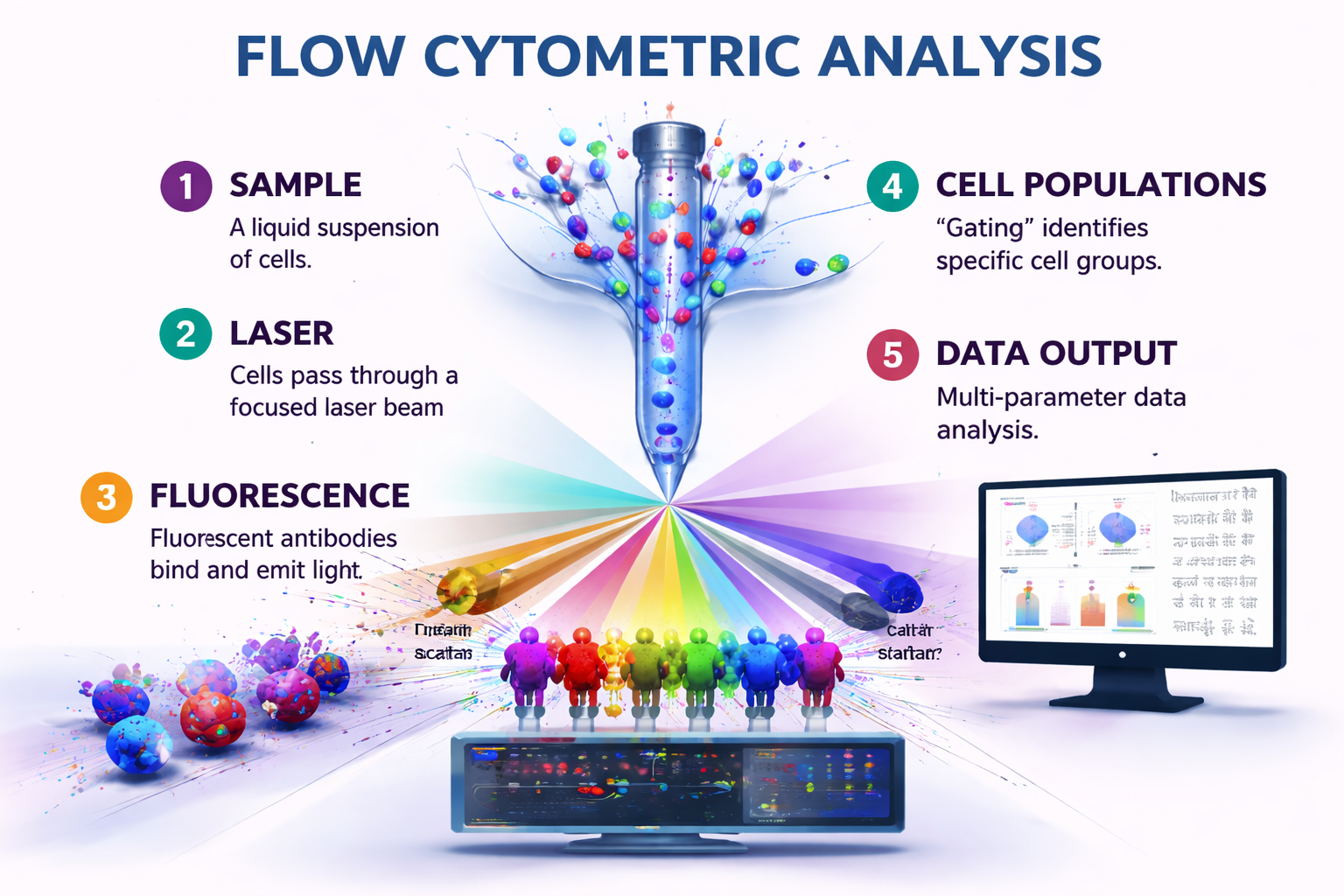
The main idea behind flow cytometric analysis is to measure the physical and chemical properties of cells (or particles) that are floating in a liquid. Think of it as a census of cells that not only counts the number of people but also keeps track of their jobs, clothes, and other small data. This flow cytometric skill has gone from being a niche tool to being very important in immunology, cancer research, stem cell biology, and drug discovery. This article goes into great detail about how flow cytometry analysis works, why it is so important, and how it is changing medicine for the better.
The Core Principle: Interrogation in a Stream
Flow cytometric analysis is brilliant because it all works so beautifully together. It has parts that work with fluids, light, and electronics.
- The Flow: A liquid suspension of cells is injected into a fast-moving stream of sheath fluid. This hydrodynamic focusing makes the cells line up in a single file, like cars going into a narrow tunnel. This is really critical for looking at each cell by itself.
- The Interrogation Point: The aligned cells then go through the middle of the instrument, which is a focused beam of light, usually from one or more lasers. This is the time to find out the truth.
- Scattering And Fluorescence:- When a cell crosses the laser beam, two key things happen.
- Light Scatter: The cell physically bends the light from the laser. Forward Scatter (FSC) is roughly connected to the size of the cells; larger cells scatter more light in front of them. Side Scatter (SSC) tells you how detailed and complicated a cell is on the inside. A granulocyte with a lot of granules, for instance, will have a high SSC.
- Fluorescence Emission: This is the part of flow cytometric analysis that really stands out. Before they are employed, cells are frequently covered in fluorescent antibodies or dyes that stick to specific targets. An antibody with a green fluorescent dye (like FITC) on it might bind to a protein called CD4 that is on the surface of a T-cell. The laser hits this dye, which absorbs the light and sends it back out at a longer, more particular wavelength (in this case, green). Modern technology can pick up more than one fluorescent colour at a time, from violet to far red.
4.Detection and Data Conversion: High-tech lenses and mirrors collect the light and fluorescence that is spread out and transfer it to a set of photomultiplier tubes (PMTs) and other detectors. These detectors turn light signals into electrical signals. We turn the height and area of each pulse into numbers, which gives each cell a number for each parameter.
Instead of a vague average, you can obtain a comprehensive, multi-dimensional dataset for thousands to millions of individual cells in only a few minutes. Flow cytometry analysis can look at single cells, which is what sets it apart from bulk approaches.
The Art and Science of Panel Design

The most important intellectual effort happens at the bench before a sample ever gets to the machine: panel design. This is the careful choice of fluorescent markers (antibodies or dyes) that will be utilised to make a detailed picture of the cell group you want to study.
A researcher investigating the immune response in cancer may develop a panel to simultaneously identify and characterise numerous immune cell types. They might have antibodies that target:
- CD45 is a pan-leukocyte marker that is used to find immune cells.
- CD3 (T-cells).
- CD4 and CD8 are helper and cytotoxic T-cells.
- CD19 (B-cells).
- CD56 (Natural Killer cells).
- PD-1 or CD25 are examples of activation markers.
- Intracellular markers like cytokines (for example, IFN-gamma) can be found. After making the cells permeable.
This multi-parameter capacity, which today typically exceeds 20–30 colours on sophisticated spectral cytometers, enables highly detailed flow cytometric analysis. It helps scientists go beyond just identifying things to figuring out how they work, how they communicate with each other, and how cells are related to each other in more complicated ways. Modern flow cytometric panels are like a master painter’s palette, allowing you to make cellular landscapes that are quite intricate.
From Diagnosis to Discovery: Applications That Define Modern Research
Flow cytometric analysis is very helpful and is an important part of both research and treatment pipelines.
- Immunology and Immunophenotyping:- Flow cytometry is especially good at. It is the best way to find out what kinds of immune cells are there, keep an eye on immunological problems (such the progression of HIV through CD4 levels), and find leukaemias and lymphomas by looking for aberrant surface protein expression on blood cancers.
- Cell Cycle and Proliferation: Scientists can employ dyes like Propidium Iodide (PI) that stick to DNA to look at a group of cells and see how many are in the G1, S, or G2/M phase of the cell cycle. Stopping the cell cycle is a very important approach to find out how effectively a drug works. This is why it is so important for cancer research and treatment development.
- Cell Health and Apoptosis: Flow cytometry can tell the difference between cells that are living, dead, or dying. You can t ell that cells are dying early by looking at how the cell membrane changes (Annexin V binding) and how healthy the mitochondria are.
- Intracellular Cytokine & Signaling Analysis: By fixing and permeabilizing cells, scientists may use flow cytometry to see within them. This is known as intracellular cytokine and signalling analysis. You can use this to look at proteins that have been phosphorylated (which are important in signalling pathways like STAT or MAPK) or cytokines that some cells make. This connects identifying the surface to how it works inside.
- Stem Cell Research: Flow cytometry analysis utilising specific surface markers (such as CD34 and CD133) is essential for identifying and isolating rare types of stem cells, including haematopoietic stem cells (HSCs). This is directly used in bone marrow transplants and therapy that helps the body heal itself.
- High- Throughput Screening (HTS): When flow cytometers are used with automated sample loaders, they may screen hundreds of chemical compounds or genetic changes in a single day. They can find “hits” that change a cell’s phenotypic in the way you want.
- Genome Editing Verification: People typically use flow cytometry to quickly check how well gene knockout worked after CRISPR-Cas9 editing by looking at the loss of a protein on the cell’s surface or inside it.
The Advent of Spectral Flow Cytometry: A Paradigm Shift
Spectral flow cytometry is a big step forward in flow cytometric analysis technology because it is now widely used. In “conventional” cytometry, optical filters send certain wavelengths of light to certain detectors. This works, but it has some problems, especially as there are more colours. This is because the signal overlap spreads out, which requires complicated math to fix.
Spectral flow cytometry works in a different way. It gets the whole emission spectrum of each fluorochrome at all times. It doesn’t just get a few data points per cell per channel; it gets hundreds of data points across a range of wavelengths. Powerful algorithms then break down these entire spectra to find the unique “fingerprint” of each fluorochrome in the cell.
The benefits are very clear:
- Multiplexing has gotten a lot better: It makes it easier to use additional fluorochromes with overlapping spectra.
- Less Need for Compensation: The unmixing algorithms do most of the work of separating the signals.
- Better Sensitivity: It can better tell the difference between weak signals and background noise and autofluorescence.
- Flexibility and Discovery: It lets you utilise dyes that aren’t usually used and can even find unknown fluorescence signals after they have been acquired.
This change makes flow cytometric analysis more powerful, easier to use for complicated research, and able to find biological changes that are more subtle than ever before.
The Data Deluge: Analysis as the New Frontier

Getting the data is only half of what you need to do. A complex flow cytometric analysis with 30 parameters can provide you billions of data points. The conventional method of manual “gating”—successively defining segments on 2D plots—remains essential yet is fundamentally subjective and complicates the study of high-dimensional data.
This has made it possible to use computers in more advanced ways:
- Automated Clustering Algorithms: Tools like t-SNE, UMAP, and FlowSOM can show high-dimensional flow cytometry data in two dimensions. This could help you uncover natural cell clusters that you might miss if you do it by hand.
- Finding populations: These algorithms are fair since they let the data show what populations are present. This helps find new, strange, or transitional cell states.
- Reproducibility: Computational pipelines can let labs analyse data in a more consistent and reproducible way, which is particularly significant in modern science.
So, a modern flow cytometric scientist needs to know how to use bioinformatics tools as well as pipettes. This is the point when modern technologies and excellent data science come together to make flow cytometry analysis better in the future.
Looking Ahead: The Future Flow
Flow cytometric analysis is not a technology that stays the same. It is always changing.
- Mass Cytometry (CyTOF): This method uses mass spectrometry to find heavy metal isotopes instead of fluorescent dyes. It gets rid of all spectral overlap, which lets you measure more than 40 parameters at once, taking multiplexing to new heights.
- Imaging Flow Cytometry: This new technology combines the high-throughput, quantitative power of flow cytometry with the visual morphology of microscopy. It takes several high-resolution pictures of each cell that goes past it. This lets scientists see where the fluorescent signal is in the cell. Is it on the membrane, in the nucleus, or in small vesicles?
- Single-Cell Sequencing Integration: The final merging. Using a flow cytometer, you can sort cells of interest live and put them directly into plates for single-cell RNA sequencing (scRNA-seq). This is a potent combination: flow cytometry gives you protein-level data and lets you sort live cells, while scRNA-seq gives you a thorough transcriptome profile. This method of combining multiple types of data at the level of a single cell is at the forefront of biomedical research.
Conclusion: An Indispensable Lens on Biology
Flow cytometric analysis has shown to be one of the most useful and powerful tools in biomedicine. It is used for anything from diagnosing blood malignancies to figuring out how the immune system works. It gives us a view that is both wide and narrow, letting us look at large groups of cells without losing sight of the single cell.
A cell only travels across the laser beam of a flow cytometer for a very short time, only a few microseconds. But the knowledge we obtain from that trip can help us understand how a terrible disease works, find the target of a promising new treatment, or find a rare stem cell that could help. The technology will only become more important as it develops through spectrum unmixing, computational analytics, and interaction with other omics platforms.Flow cytometric analysis is a basic language that current biomedical research uses and will continue to use. It lets us read the complex story of life, one brilliant, flashing cell at a time.